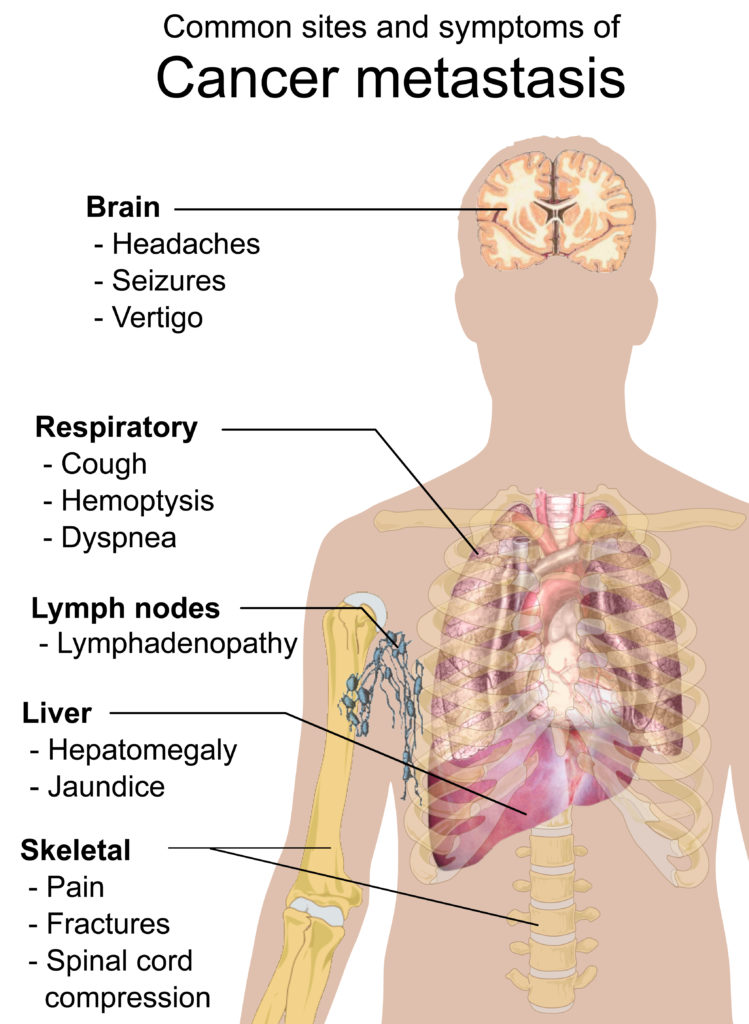
Cancer patients whose disease has spread to the brain or the central nervous system (CNS) are traditionally excluded from clinical trials of new anti-cancer therapies.
However new guidelines from an international, multidisciplinary group published in the journal Lancet Oncology describe how to most appropriately address cancer patients with CNS involvement within clinical trials of anti-cancer drugs.
“Two major situations needed to be optimized. Firstly, as we’ve actually started to see some new anti-cancer drugs working well inside the brain, we needed to find a way to appropriately include these patients in clinical trials so that the trials could accurately capture that benefit. And secondly, for drugs that would be unlikely to work in the brain, we needed to limit risks to patients and to the drug development process,” says D. Ross Camidge, Joyce Zeff Chair in Lung Cancer Research at the University of Colorado Cancer Center, director of Thoracic Oncology at the CU School of Medicine and the lead author of the trial design guidelines.
The expert working group which developed these guidelines, called the Response Assessment in Neuro-Oncology – Brain Metastases (RANO-BM) group, also includes researchers from Dana-Farber Cancer Institute, City of Hope National Medical Center, Cleveland Clinic, University of Heidelberg in Germany, University of California at San Francisco, Queen’s University in Canada, University of Groningen and Erasmus University Medical Center in the Netherlands, University of Turin in Italy, Massachusetts General Hospital, University of Virginia, M.D. Anderson Cancer Center, and Columbia University Medical Center.
“Historically, patients with brain metastases were excluded from the majority of systemic therapy trials for a number of reasons, including the misperception that they are poor clinical trial candidates. However, many studies show that select patients with brain metastases can safely enroll on clinical trials, without harm to the patient or to the drug development process,” says Eudocia Lee, assistant professor of Neurology at the Center for Neuro-Oncology at the Dana-Farber Cancer Institute and co-lead author of the guidelines.
The guidelines adopt a pragmatic approach, suggesting one of three specific strategies based on initial understanding of a drug’s possible activity in the brain. First, when a new drug is considered very unlikely to have activity in the brain, patients with stable CNS disease should be permitted, while those with active CNS disease should be excluded from trials of systemic therapy.
Second, if there is some initial evidence that a drug may have activity in the brain, the guidelines propose including patients with both stable and active CNS disease in a way that will capture data defining a drug’s activity in the brain separate from its activity in the rest of the body.
Third, when it’s unclear whether a drug may have activity in the brain (as is often the case at the start of any new drug development process), the guidelines suggest including a dedicated cohort of patients with brain metastases very early in drug development to generate the data that would allow trial designers to adopt one of the other two trial designs.