The Drugs Controller General (India) has granted permission to an Indian firm to conduct Phase-I clinical trial or a potential candidate for a zika vaccine. Zika is an infection spread by the Aedes egypti mosquito. Phase I trials ascertain the safety, tolerability and physiological action of a compound inside the body.
Union Minister of State for Health and Family Welfare Ashwini Kumar Choubey told the Lok Sabha on Monday that an Indian firm had submitted an application to the drugs controller. “The application was on the Zika virus vaccine, along with non-clinical (Animal) Toxicity data, claiming 100 per cent efficacy in animals for grant of permission to conduct the Phase-I clinical trial. Based on the evaluation of application, in consultation with the Experts Committee, the DCG (I) has granted permission to conduct the Phase-I clinical trial,” he said in a written reply to a question.
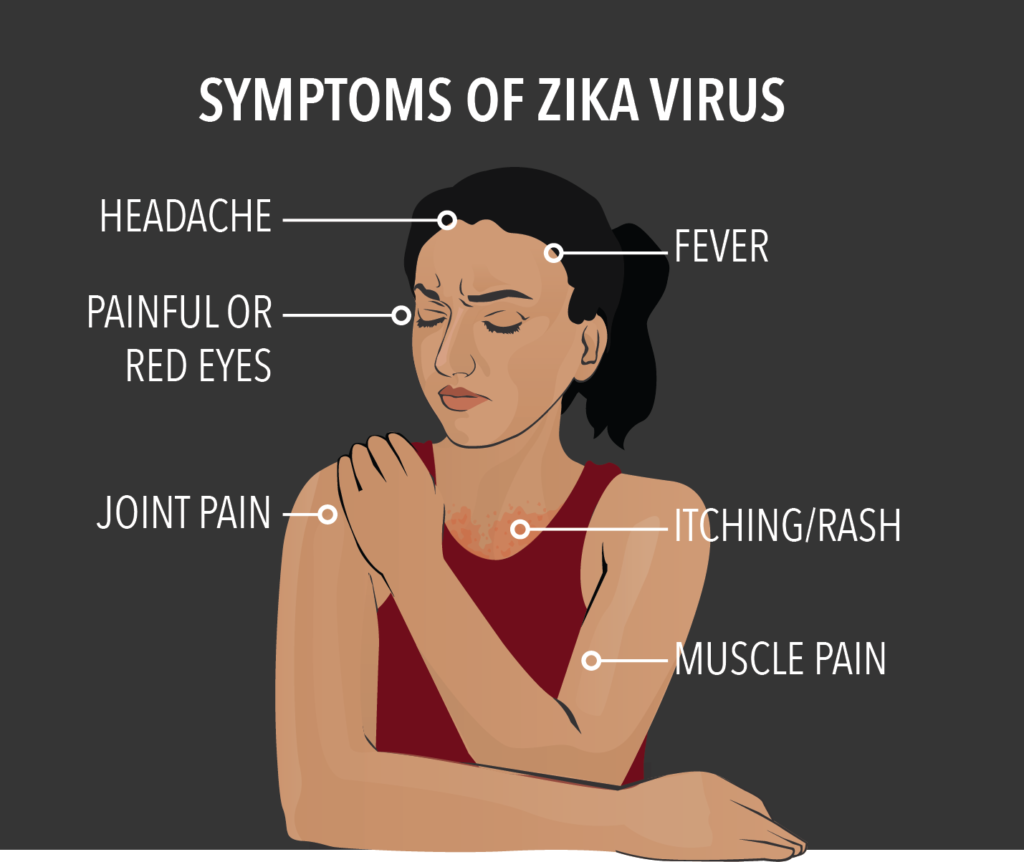
Anupriya Patel, also Union Minister of State for Health and Family Welfare, said in another written reply that the World Health Organisation (WHO) had declared the Zika virus disease to be a Public Health Emergency of International Concern (PHEIC) on February 1, 2016, following the Zika outbreak in Brazil and other Latin countries and its association with birth defects (microcephaly) in new borns. Microcephaly is a condition in which children are born with undersized heads; there are usually accompanying brain deficiencies.
“Further, the WHO declared that it ceased to be a Public Health Emergency on November 18, 2016,” Patel said. She added that the ministry has a three-pronged action plan on combating the disease, and there are 27 laboratories in the government sector that can test the Zika virus disease.
HOW ZIKA SPREADS
1. A mosquito bites a person infected with Zika virus
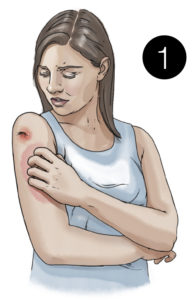
2. The mosquito becomes infected

3. The infected mosquito bites a person and infects them with Zika

4. Other mosquitoes bite that person and become infected
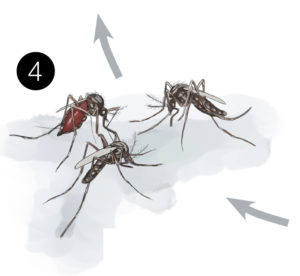
5. More members of the community become infected when they are bitten by those infected mosquitoes
Source: CDC